Soft matter
Water-like liquids as short-chain polymers
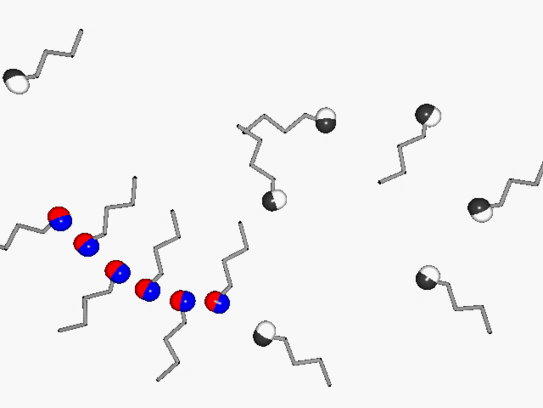
Hydrogen bonds determine the structure and dynamics of numerous liquids and solids. These bonds are not only of central importance for many biophysical processes, but also for the microwave absorption of water, which can form a complex supramolecular structure. This is responsible for the appearance of a relatively narrow but very intense absorption band called the Debye peak. Other Debye fluids we have studied that share this characteristic with water include amides - which contain peptide bonds - and monohydroxy alcohols. The animation schematically illustrates their transient structure formation, in which the short-chain polymer chain formed from OH groups is constantly reinventing itself.